Aspartame and Cancer Link: Investigating the Scientific Rationale for the Classification
More scientific studies needed to establish the link between aspartame and cancer risk
Dear Readers,
Welcome to Science Simplified, a newsletter which Unlocks the Wonders of Science, One Simplified Concept at a Time!
In this edition, we will embark on an intriguing journey into the bittersweet world of aspartame. As a chemical sweetener, aspartame has captured both attention and anxiety for four decades since its inception. Recently, the World Health Organization (WHO) classified it as a potential carcinogen, rekindling concerns and sparkling crucial questions about its safety. This marks the first time such a prominent body has publicly addressed the effects on aspartame, which has been a contentious ingredient for decades.
In this comprehensive article, we aim to address all the lingering concerns you may have about aspartame, shedding light on the scientific facts behind the controversy. We hope by the end of this article, you will have a comprehensive understanding of the current scientific knowledge surrounding aspartame, empowering you to make informed decisions about its place in your diet and lifestyle.
Thank you for subscribing to Science Simplified. Kindly consider upgrading to a paid subscription to support our newsletter.
By doing so, not only will you gain access to additional benefits, but you will also contribute to the continued production of informative content that can make a positive impact on the lives of our readers. Thank you so much for your support!
What is Aspartame ?
In the realm of sugar substitutes, Aspartame (known as E951 in the European Union) has taken center stage, captivating millions as a low- or zero-calorie alternative to Sugar. However, the story behind its rise and the controversies that surround it reveal a complex and intriguing tale.
Aspartame's journey began in the mid-1960s when a chemist named James Schlatter accidentally discovered its sweet taste while working on an anti-ulcer drug. Schlatter noticed the sweet properties of the compound as he licked his finger during a routine laboratory experiment. This serendipitous event sparked the curiosity of scientists and set the stage for the development of one of the most widely used artificial sweeteners.
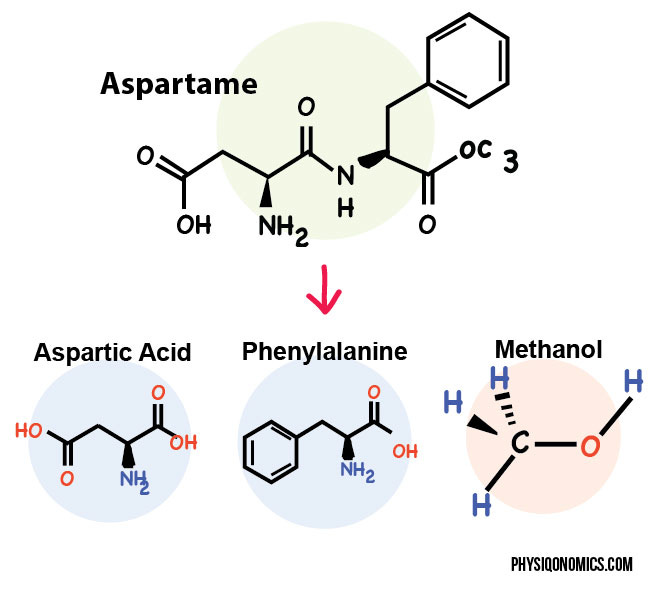
Metabolism of Aspartame: At its core, aspartame is a dipeptide sweetener composed of two amino acids: aspartic acid and phenylalanine, linked together. Its chemical formula is C14H18N2O5. Once aspartame is ingested, it encounters hydrolysis in the small intestine, where water molecules break it down. This process splits aspartame into its two amino acids. These amino acids are naturally occurring and are found in many protein-containing foods. A small amount of methanol is also formed as aspartame is metabolised. Methanol is further broken down into formaldehyde and then into formic acid. Higher levels of formaldehyde are harmful to body and potential carcinogen. It's worth mentioning that aspartame metabolism does not lead to the formation of harmful levels of methanol and formaldehyde.
The unique combination of these amino acids contributes to the intense sweetness of aspartame, which is approximately 200 times sweeter than table sugar (sucrose).
The saga of controversies began before its approval
The controversies surrounding aspartame began even before its approval for use as a sweetener. From the early stages of its development, aspartame faced scrutiny and debates over its safety and potential health effects. Aspartame was discovered in 1965, and by 1974, the U.S. Food and Drug Administration (FDA) had approved its use in dry foods. However, due to lingering safety concerns, the approval process faced delays, and it wasn't until 1981 that the FDA granted approval for its use in carbonated beverages. The company involved in the approval process was G.D. Searle & Company. In 1984, this company was bought by Monsanto.
Prior to the official approval, there were some early studies conducted on rats which raised concerns linking aspartame to cancer risk. These concerns were largely based on the fact that aspartame can metabolise into menthol, which may convert into formaldehyde, a potential carcinogen. Additionally, in 1977, an FDA scientist named Dr Adrian Gross reviewed these initial studies and published his findings in Bressler report. In Bressler Report, Dr Gross raised serious concerns about the safety of aspartame and recommended against the approval of aspartame.
It's important to highlight that these initial studies linking aspartame to cancer risk in rats were widely criticised for various methodological issues. One major concern was that the doses of aspartame fed to the rats were higher that the recommended human intake levels. Additionally, rat metabolism differs from human metabolism, making direct comparisons challenging. Eventually, the FDA concluded that aspartame was safe for consumption within the acceptable daily intake limits established by regulatory authorities.
Aspartame started Sugar-Free, Zero-Calorie Movement
Aspartame was marketed under various brand names. Some of the well-known aspartame-based products include NutraSweet, Equal, and Canderel, among others. These brand names became synonymous with the Sugar-Free Movement, offering consumers the promise of sweetness without the guilt of excess calories. This occurred during an era when sugar was labeled as the primary culprit behind weight gain, obesity-related metabolic disorders, such as diabetes and cardiovascular diseases, cavities and tooth decay.
The Diet Soda Revolution: One of the significant milestones in the aspartame saga was its incorporation into carbonated beverages, particularly diet sodas. With concerns over the health risks associated with sugar consumption, aspartame became the go-to ingredient for companies seeking to cater to health-conscious consumers. Iconic brands like Diet Coke, Coke Zero, and Diet Pepsi gained popularity and became staples in the diet soda market. The allure of sugar-free options and the desire to maintain a svelte figure in line with societal beauty standards propelled the rise of aspartame-laden beverages.
How many products use aspartame? Aspartame is used in more than 6,000 products, besides Diet Coke and Diet Pepsi, it can be found in Kool Aid, Crystal Light, Tango and other artificially sweetened drinks; sugar-free Jell-O products; Trident, Dentyne and most other brands of sugar-free gum; sugar-free hard candies; low- or no-sugar sweet condiments such as ketchups and dressings; children’s medicines and vitamins.

Regulatory Authorities validates Aspartame as SAFE
Aspartame, one of the most studied food additives in the human food supply, has consistently received approval as a safe sweetener for human consumption. Over 100 regulatory agencies worldwide, including the US FDA, the UK Food Standards Agency, the European Food Safety Authority (EMSA), Health Canada, and Food Standards Australia New Zealand, have thoroughly evaluated the scientific evidence and endorsed the safety of aspartame in food and beverages.
In 2013, EFSA published its “full risk assessment” of aspartame, which concluded that aspartame and its breakdown products were safe for the general population. Reaffirming this confidence, the Joint Expert Committee on Food Additives (JECFA) upheld the acceptable daily intake (ADI) of 40 mg/kg body weight as of July 15, 2023.
What does the ADI mean for consumers? With a typical diet soft drink containing approximately 200-300 mg of aspartame, an adult weighing 70 kg would need to consume more than 9-14 cans per day to surpass the acceptable daily intake, assuming no additional intake from other food sources.
WHO classifies aspartame ‘POSSIBLE CARCINOGEN’
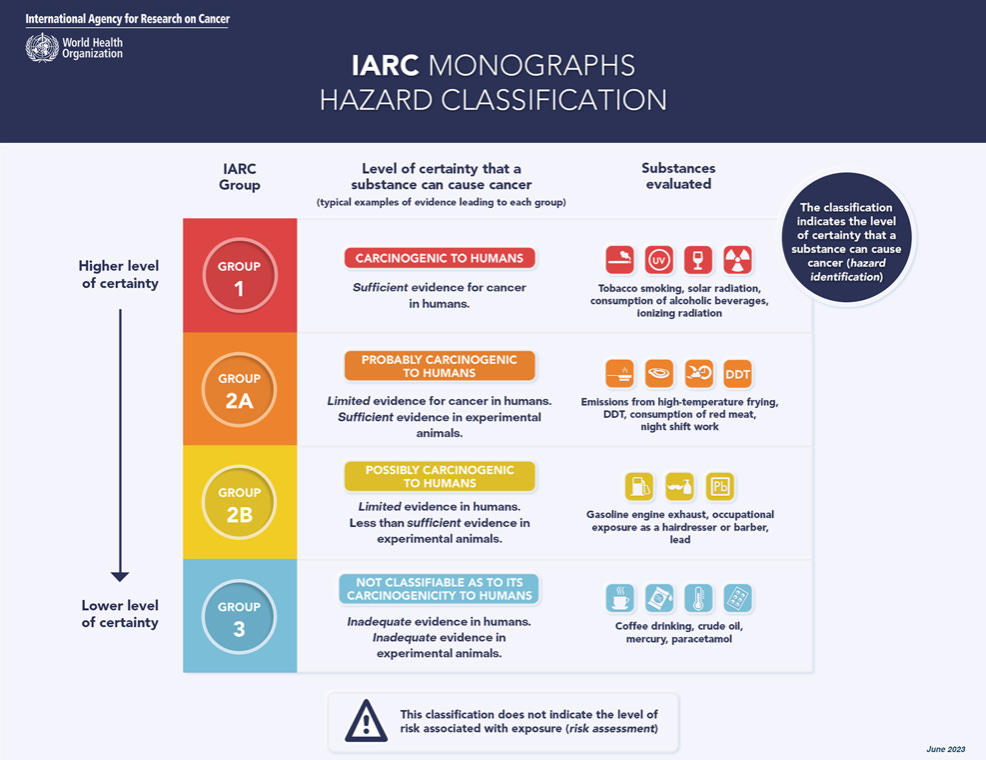
On July 14, 2023, a combined assessment report raised some concerns about the possible carcinogenicity of aspartame, citing "limited evidence" for carcinogenicity in humans. Notably, the International Agency for Research on Cancer (IARC) categorized aspartame as "possibly carcinogenic to humans" (IARC Group 2B).
However, it's crucial to recognize that the scientific consensus among regulatory agencies, such as the FDA and the EFSA, is that the available evidence does not support a definitive carcinogenic effect of aspartame in humans when consumed within the approved daily intake levels. These regulatory bodies have conducted thorough reviews of multiple studies and have consistently deemed aspartame to be safe for human consumption.
In the same report, the IARC also classified other substances, such as extracts of aloe vera, traditional Asian pickled vegetables, certain vehicle fuels, and some chemicals used in dry cleaning, carpentry, and printing, as "possibly carcinogenic." Additionally, the IARC has classified red meat as "probably carcinogenic" and processed meat as "carcinogenic" based on their potential association with cancer.
The Scientific Basis for Aspartame's Possible Carcinogenicity
The IARC classified aspartame as "possibly carcinogenic to humans" (Group 2B) based on limited evidence for cancer in humans, specifically hepatocellular carcinoma, a type of liver cancer. There was also limited evidence for cancer in experimental animals and limited evidence related to the possible mechanisms for causing cancer.
The IARC's classification system is based on the evaluation of scientific evidence regarding the potential carcinogenicity of various substances. The classification of aspartame in Group 2B signifies that there is limited evidence suggesting a possible association between aspartame and cancer in humans, but the overall evidence is not sufficient to establish a definite causal relationship.
A. Scientific Studies revealing link of aspartame with Increased Risk of Liver Cancer
The IARC's evaluation considered various studies, including both animal and human research, to determine the potential carcinogenicity of aspartame. The limited evidence for hepatocellular carcinoma in humans comes from three studies that examined the consumption of artificially sweetened beverages.
A Study published in 2014, followed 477,206 participants in 10 European countries for more than 11 years and showed that the consumption of sweetened soft drinks, including those containing aspartame, was associated with increased risk of a type of liver cancer called hepatocellular carcinoma.
A 2022 US-based study showed that consumption of artificially sweetened drinks was associated with liver cancer in people with diabetes.
The third study, involving 934,777 people in the US from 1982 to 2016, found a higher risk of pancreatic cancer in men and women consuming artificially sweetened beverages.
Limitation of above studies
Association, not causation: These studies establishes an association between the consumption of sweetened soft drinks and an increased risk of hepatocellular carcinoma, but it does not prove a direct cause-and-effect relationship.
Potential confounding factors: Other variables, such as overall diet, lifestyle, and underlying health conditions, could influence the observed association but may not have been fully accounted for in the study design.
Self-reporting bias: The reliance on self-reported dietary information could introduce inaccuracies and recall bias, potentially affecting the validity of the results.
Reverse causality: The possibility of reverse causality exists, where underlying health conditions or other factors may influence beverage consumption patterns rather than the beverages causing the health outcomes.
Limited generalizability: The study focused on a specific population in European countries, and the findings may not necessarily apply universally.
B. Scientific Studies highlighting the potential role of aspartame as carcinogen
This study found that among 102,865 adults in France, people who consumed higher amounts of aspartame (but less than the recommended ADI) had an increased risk of breast cancer and obesity-related cancers. This study does not have enough statistical power to investigate liver cancer for the moment.
Series of large-scale toxicological studies of the possible carcinogenicity of aspartame were conducted in 2006 and 2007 by Ramazzini Institute (RI), an independent, not-for-profit research laboratory in Bologna, Italy. However, it's worth noting that the study's findings have been the subject of significant scientific debate and criticism due to several methodological limitations and inconsistencies with other studies. The EFSA made the unsubstantiated claim that the RI’s animal colony was poorly managed and that the experimental animals were subject to uncontrolled infections. Other scientists speculated that lesions on the lung diagnosed as cancers by RI might have been inflammatory lesions caused by bacterial infections.
In a 2021 review of the Ramazzini Institute data, researchers at Boston College validated the conclusions of the studies. A 2023 study by the Ramazzini Institute in the Annals of Global Health reports on all the detailed haemolymphoreticular neoplasias (HLRN) diagnoses of the Ramazzini Institute rats and mice studies on aspartame and the related statistics.
Limitations of above Studies:
The agency concluded that the results of the Ramazzini study did not provide sufficient evidence to alter their previous determination that aspartame is safe for consumption within approved limits. The FDA's evaluation took into consideration the study's limitations and inconsistencies, as well as the broader body of research on aspartame.
C. Epidemiological Studies
Epidemiological studies are scientific investigations that focus on understanding the distribution and determinants of health-related events, diseases, or conditions in specific populations. Till now two epidemiological studies have been conducted of aspartame-exposed populations. Let's examine each study in more detail:
US National Cancer Institute Study: In 2006, the US National Cancer Institute conducted a large-scale study involving a significant population of middle-aged Americans. The objective of this study was to evaluate the potential carcinogenic effects of aspartame. The findings of this study did not indicate any evidence of a carcinogenic effect associated with aspartame consumption. It suggested that, within the studied population, aspartame did not increase the risk of developing cancer. However, it's important to note that this study focused on middle-aged Americans and may not necessarily be representative of all demographic groups.
Harvard Nurses Health Study: Another epidemiological study, conducted within the framework of the Harvard Nurses Health Study, specifically assessed the relationship between aspartame consumption and non-Hodgkin lymphoma (NHL) risk. This study meticulously followed a population of nurses and carefully evaluated their exposures. The study found a significantly elevated risk of NHL in males who consumed one or more servings of soda per day. It is worth mentioning that this study did not specifically examine aspartame as an isolated factor, but rather assessed the association between soda consumption (which may contain aspartame) and NHL risk. This association suggests a potential link between soda consumption and NHL in males, but the study did not exclusively attribute it to aspartame itself.
Let’s Summarise!
The scientific studies linking aspartame consumption with higher risk of cancers have limitations. It is crucial to avoid overgeneralization and to recognize the need for further research, including well-designed prospective studies and controlled trials, to establish more definitive conclusions regarding the relationship between aspartame consumption and cancer risk.
Additionally, it is worth emphasising that the majority of studies investigating aspartame and carcinogenicity have been conducted on animals, particularly rodents. High-dose studies in animals have shown an increased risk of certain cancers, but these doses are significantly higher than typical human consumption levels. Extrapolating the results of animal studies to humans is challenging and requires careful interpretation.
Furthermore, the mechanism by which aspartame could potentially act as a carcinogen is still a subject of ongoing scientific research and debate. While some studies have suggested possible mechanisms, it is important to note that the evidence is not conclusive, and further research is needed to establish a definitive link.
Concluding Note: Navigating the complex world of Artificial Sweeteners
The realm of artificial sweeteners demands a thoughtful approach, combining scientific knowledge, individual considerations, and moderation. Aspartame, with its remarkable sweetness and storied past, serves as a poignant reminder that our understanding of sweeteners and their impact on health remains an evolving journey.
Before classifying aspartame as a ‘possible carcinogen’, the WHO also issued a strong recommendation against common non-saccharide sweeteners (NSS), including aspartame, for weight management and reducing the risk of non-communicable diseases. Their stance was based on a systematic review of the available evidence, which revealed that the consumption of NSS does not yield any long-term benefits in reducing body fat for both adults and children.
Additionally, WHO also raised concerns about potential undesirable effects stemming from prolonged use of aspartame and other non-saccharide sweeteners. Among these concerns were indications of an increased risk of type 2 diabetes, cardiovascular diseases, and even mortality in adults. The seemingly "healthy" appeal of zero-calorie, sugar-free products now comes into question, as it appears they might not be as beneficial as once believed.
As you consider reaching for that diet soda or sugar-free snack, reflect on the intriguing tale of aspartame and make an informed choice. Perhaps, the true culprits lie elsewhere - carbonated drinks or calorie-laden snacks disguised as "sugar-free" and "healthy" options. Let wisdom guide your decisions, and remember that striking a balance and being mindful of your overall dietary choices is key to promoting a healthful lifestyle.
Please Support Science Simplified!
Lastly, we kindly invite readers to consider upgrading to a paid subscription to support our newsletter. By doing so, not only will you gain access to additional benefits, but you will also contribute to the continued production of informative content that can make a positive impact on the lives of our readers.